If you’ve heard of Ozempic, you know it’s basically the hottest drug on the planet right now. Originally approved for Type 2 diabetes, its weight-loss side effects made it a global phenomenon. Novo Nordisk, the Danish pharma giant behind it, turned Ozempic into a blockbuster worth tens of billions.
But what happens when a cheaper Indian version enters the market — and the name sounds suspiciously familiar?
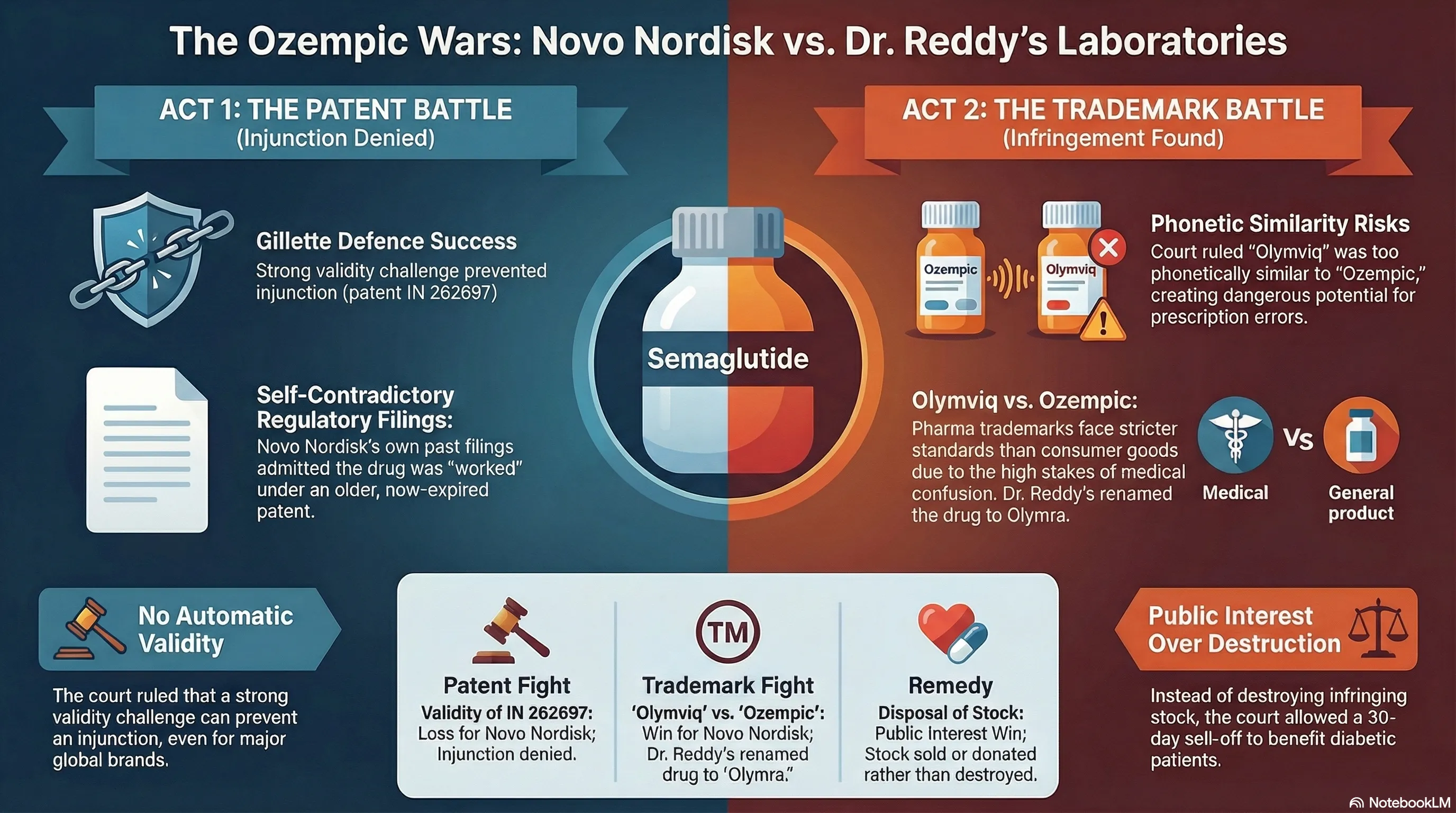
That’s the story of Novo Nordisk vs Dr Reddy’s Laboratories — a legal saga that played out in the Delhi High Court across two separate battles: one over a patent, and one over a brand name. Both went against Dr Reddy’s. But the court’s reasoning in each case is worth understanding, because it reveals something important about how Indian courts balance IP rights with public interest.
First, a quick explainer: what’s semaglutide?
Semaglutide is the active ingredient in Ozempic and Wegovy. It belongs to a class of drugs called GLP-1 receptor agonists — they help control blood sugar and suppress appetite. Novo Nordisk spent years developing it, and holds patents that protected their exclusivity in markets across the world.
In India, their key patent (IN 262697) was set to expire in March 2026. Once it did, any generic manufacturer could legally make and sell semaglutide. Dr Reddy’s, one of India’s largest pharmaceutical companies, was ready and waiting.
Act 1: The Patent Fight (December 2025)
Before the patent expired, Novo Nordisk noticed something troubling: Dr Reddy’s had already started manufacturing semaglutide in April 2025 — months before the patent lapsed.
Novo Nordisk filed for an interim injunction to stop them.
Dr Reddy’s hit back with a clever defence called the Gillette Defence — a legal argument that essentially says: “We’re not infringing your patent because your patent is invalid anyway.”
Their argument? Semaglutide was already covered by an earlier, broader patent (called the Genus Patent, IN 275964) that had expired in September 2024. That earlier patent, they said, already disclosed the key building blocks of semaglutide — the specific amino acid substitution that makes semaglutide what it is was already described in the older patent’s claims.
Here’s where it gets interesting: Novo Nordisk’s own paperwork worked against them. The company had filed annual Form 27 returns (a regulatory requirement in India to declare commercial working of patents), stating that their Genus Patent had been “commercially worked” in India — using semaglutide revenues to demonstrate this. The court found these declarations to be a binding admission that semaglutide was already covered by the earlier patent.
Justice Manmeet Pritam Singh Arora declined to grant the injunction. With the patent’s validity seriously in doubt, and its expiry just months away, stopping Dr Reddy’s manufacturing didn’t make sense.
Key takeaway from Act 1: In India, there’s no automatic presumption that a granted patent is valid. A strong challenge to validity can prevent an interim injunction — even from a well-resourced multinational.
Act 2: The Trademark Fight (March 2026)
The patent expired. Dr Reddy’s launched its semaglutide injection in India. Initially, they called it “Obeda” — priced significantly lower than Ozempic and Wegovy.
Then they also tried to sell it under another brand name: “Olymviq.”
Novo Nordisk immediately filed a trademark infringement suit.
Their argument was simple: say “Olymviq” out loud. Now say “Ozempic.” Sound familiar?
Justice Jyoti Singh agreed. The court found a prima facie case of phonetic similarity between the two marks. In pharmaceutical trademark cases, courts apply a stricter standard than for regular consumer goods — because when a doctor scribbles a prescription, or a pharmacist takes a telephonic order, even a slight chance of confusion can have serious consequences for a patient.
The judge put it plainly:
“According to me, there is a phonetic similarity… and in a pharma, it’s a threshold which I would not like to cross.”
The court also noted something telling: Dr Reddy’s had described “Olymviq” in its own trademark applications as “proposed to be used” — yet simultaneously claimed in court that it was already commercially launched. This inconsistency didn’t help their case.
The Twist: Public Interest Over Punishment
Novo Nordisk won the trademark case. But what happens to all the Olymviq stock already sitting in warehouses?
Typically, in trademark infringement cases, courts can order infringing goods to be destroyed. Novo Nordisk pushed for exactly this.
Dr Reddy’s pushed back — and made a practical argument: the drug is temperature-sensitive and can’t simply be relabelled. Destroying it would mean wasting functional, quality-tested medicine that diabetic patients could benefit from.
Justice Singh sided with public interest:
“In my view, it will be in public interest to permit the sale of the existing stock instead of directing its destruction.”
The court allowed Dr Reddy’s to sell existing Olymviq stock for 30 days. Any unsold stock remaining after that had to be donated to a government hospital — not destroyed.
Dr Reddy’s also undertook to rename the drug to “Olymra” going forward. Novo Nordisk’s lawyers indicated this was acceptable.
Novo Nordisk was awarded 30% of its claimed costs.
Why This Case Matters
For patients: Cheaper semaglutide is now available in India. The patent battle ensured that generic competition couldn’t be blocked. Dr Reddy’s “Obeda” launched at a fraction of Ozempic’s price — making the diabetes drug accessible to millions more Indians.
For brand owners: The trademark win shows that even in generics-friendly India, brand names matter. A phonetically similar name on a competing drug will not survive court scrutiny — especially in pharmaceuticals.
For the pharma industry: Indian courts are not afraid to question patent validity, especially when a company’s own regulatory filings contradict its litigation position. Be careful what you tell the patent office versus what you argue in court.
For IP lawyers: The “public interest in access to medicine” argument is increasingly being used to shape remedies in IP cases — not to deny rights, but to determine how those rights are enforced. Destruction of medicine isn’t a given anymore.
The Bottom Line
Novo Nordisk fought on two fronts. They lost the patent battle (their own paperwork undermined them), but won the trademark battle (Olymviq sounded too much like Ozempic). Dr Reddy’s gets to sell its cheap semaglutide — but under a different name, and with a side order of court costs.
It’s a case that sits at the intersection of innovation, access to medicines, and the power of a brand name. And it’s a preview of the kinds of IPR battles that will define the global generics industry as more blockbuster drug patents expire in the coming years.
Sources: Bar and Bench · Indian Kanoon · LawChakra · Medical Dialogues

